OMRI: Driving WA medical research and innovation into 2026 and beyond

By WA, for WA. Every breakthrough starts with a patient and a problem to solve. In 2026, the Office of Medical Research and Innovation (OMRI) is focused on accelerating ideas into outcomes—backing local teams with targeted funding, building clinical trials networks, streamlining approvals through the Central Human Research Ethics Committee, and activating innovation hotspots at Murray Street, QEII and Floreat—so homegrown ideas reach Western Australians sooner.
OMRI is WA Health’s system manager for health and medical research and innovation. Working alongside the Future Health Research and Innovation (FHRI) Fund, we connect partners across government, academia, industry and community to improve health outcomes and grow WA’s innovation economy.
Vision for a healthy and prosperous WA
OMRI supports the operations of the FHRI Fund, a long‑term, sovereign investment designed to drive health and medical research, innovation and commercialisation for the benefit of all Western Australians.
Under the FHRI Fund Strategy 2025–30 (the Strategy), our shared vision is to triple the impact of health research and innovation by 2035 through a sharper focus on WA’s unique strengths and needs.
By building a connected pathway from discovery to manufacturing, the FHRI Fund and OMRI are helping create high-value jobs in research, clinical trials, data science and advanced biomanufacturing, while positioning WA as a destination of choice for medical research and innovation.
Background
OMRI sits within the Department of Health and provides policy, funding and sector coordination, including research governance, intellectual property(IP) advice and administration of FHRI programs and initiatives.
Since inception in 2020, the FHRI Fund has grown into a $1.8 billion long‑term investment framework, with more than $282 million awardedto 810 recipients across hundreds of projects.
The Strategymarks a deliberate shift from broad capacity building to targeted, translational impact, underpinned by transparent governance and predictable funding pathways.
Who we are and what we do
OMRI’s role is to guide, inform and promote collaboration across WA’s health and medical research and innovation sector, and to administer the FHRI Fund Programs and Initiatives. Key functions include sector policy leadership, ecosystem development, research governance and clinical trials enablement. We steward communications and engagement across the sector—linking researchers, clinicians, innovators and communities—and provide a clear entry point via the WA Medical Research website.
Our role is to create a simpler, faster path from discovery to treatment so WA-made solutions can help people sooner.
Impact so far
FHRI Fund support is translating into wins for WA—national grants secured, partner co‑investment attracted, and projects progressing from discovery to patient care. The Fund is also strengthening WA’s research workforce through targeted fellowships and capability‑building initiatives. Targeted co-funding through MRIAS/MRAS has increased the competitiveness of WA applications to national programs and attracted additional partner investment.
FHRI seed commitments to WA teams convertstrong proposals into nationally competitive grants and partner investment, multiplying early support into larger real-world impact.
This sits within a broader trajectory of more than $282 million granted to 810recipients, strengthening WA’s capabilities, infrastructure and talent pipeline.
2026 priorities at a glance
“In 2026, OMRI will turn strategy into delivery, linking funding to real‑world impact and powering WA’s innovation hubs.”
The Strategy is organised around three themes that shape our priorities and programs:
-
Connect and Collaborate:fostering partnerships across health, research, and innovation communities.
-
Accelerate and Translate: boosting innovation and commercialisation in WA.
-
Foundational Confidence: strengthening the sector’s competitiveness through sustained support.
Looking ahead, 2026 will be a year of delivery and continued momentum for OMRI across a broad agenda.
Implement FHRI Fund Programs and Initiatives to activate the Strategy
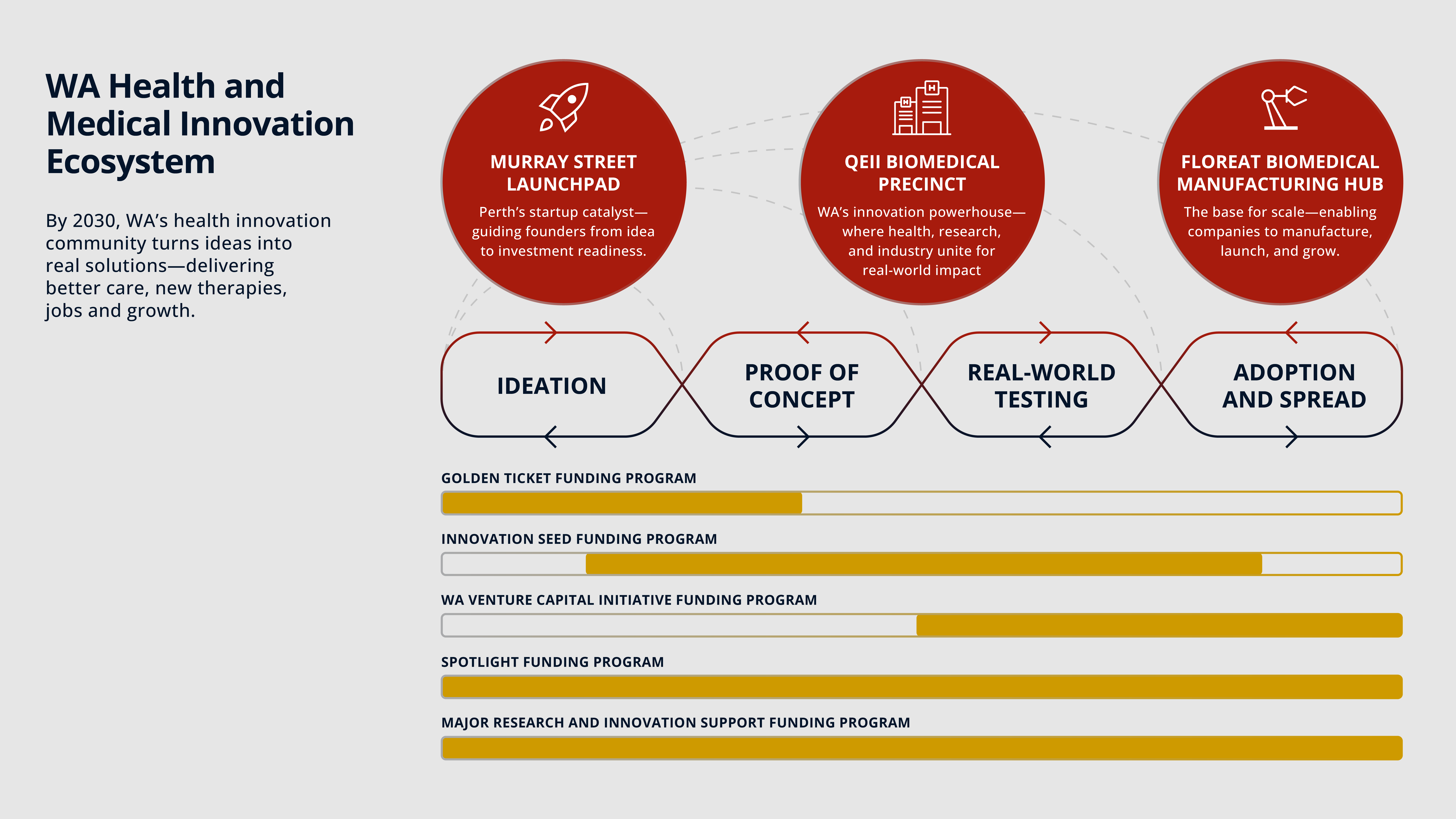
We will continue rolling out FHRI Fund programs aligned to the Strategy, including current opportunities such as Research Infrastructure Support 2026, Innovation Seed Fund 2025–26, and the Major Research and Innovation Application Support (MRIAS) rounds.
A flagship of the Strategy is the Spotlight Program, an annual, multi‑year commitment that mobilises the sector around one uniquely Western Australian health challenge. The 2025-26 Spotlight focuses on Rheumatic Heart Disease (RHD). The program provides up to $25 million in two stages: Part A (up to $1 million over 9 months) to consult and co‑design a sector‑wide Action Plan; Part B (up to $24 million over48 months) to implement the plan with partners.
Applications are now open and closing 1:00 pm (AWST), Wednesday 11 March 2026.
This work aligns with WA’s hosting of the World Heart Federation World Congress on RHD in Perth, 12-14 November 2026, offering a global platform for WA’s leadership and collaboration.
Progress the WA Clinical Trials Roadmap and National One Stop Shop (NOSS)
We’ll progress WA Clinical Trials Roadmap—embedding trials in care, growing workforce capability, and strengthening infrastructure—to expand access and improve start‑up times.
In parallel, OMRI will continue Western Australia’s transition planning to the National One Stop Shop (NOSS) for ethics and governance—a national, single‑platform approvals system.
This means trials can start up faster and more consistently across WA.
Deliver the three innovation ecosystem drivers — Murray Street, QEII and Floreat
Murray Street Launchpad (CBD): The Murray Street Launchpad is a catalyst for early‑stage health innovation, helping founders move ideas to proof‑of‑concept and investment‑readiness. The fit‑out is underway, with operations expected to commence in Q3 2026.
QEII Health & Biomedical Precinct (Nedlands): The QEII Health & Biomedical Precinct project brings together clinicians, researchers, industry and education to accelerate translation through deeper integration and collaboration. Governance and planning are progressing, supported by program funding of $1.8 million.
Floreat Biomedical Manufacturing Hub (Floreat): The Floreat Biomedical Manufacturing Hub is the State’s scale‑up anchor, enabling regulatory readiness and local manufacturing so innovations move from testing to adoption. Scoping continues under a $50 million commitment.
Expand and improve the WA Health Central Human Research Ethics Committee
We’ll consolidate and scale the WA Health Central Human Research Ethics Committee (National Health and Medical Research Council certified on 17 Oct 2025 and endorsed to participate in National Mutual Acceptance), continuing the move to a single, consistent ethics pathway that reduces duplication and speeds up approvals for researchers.
Stay connected
Big news is on the way across funding, trials, ethics and precinct activity.
How to stay in the loop:
-
Visit the FHRI website to explore current opportunities and sign up for updates.
-
Visit the Medical Research website and subscribe to our Inspireenewsletter.
-
Follow us on LinkedIn.
-
Watch recordings of past events on YouTube.
A note on language and acknowledgement
We acknowledge the Traditional Custodians of Western Australia and pay our respects to Elders, past and present. Our work—particularly through the Spotlight on RHD—emphasises collaboration with Aboriginal leaders and Aboriginal Community Controlled Health Organisations to support co‑designed, community‑led solutions.